Spinal cord injury (SCI) involves primary trauma, followed by a wave of “secondary damage” - an acute process involving radiating inflammation and cellular stress, killing neurons that survived initial impact. At the Gaudet Lab, we are searching for molecular modulators that control this process, hoping to shift inflammation to benefit tissue survival and functional recovery.
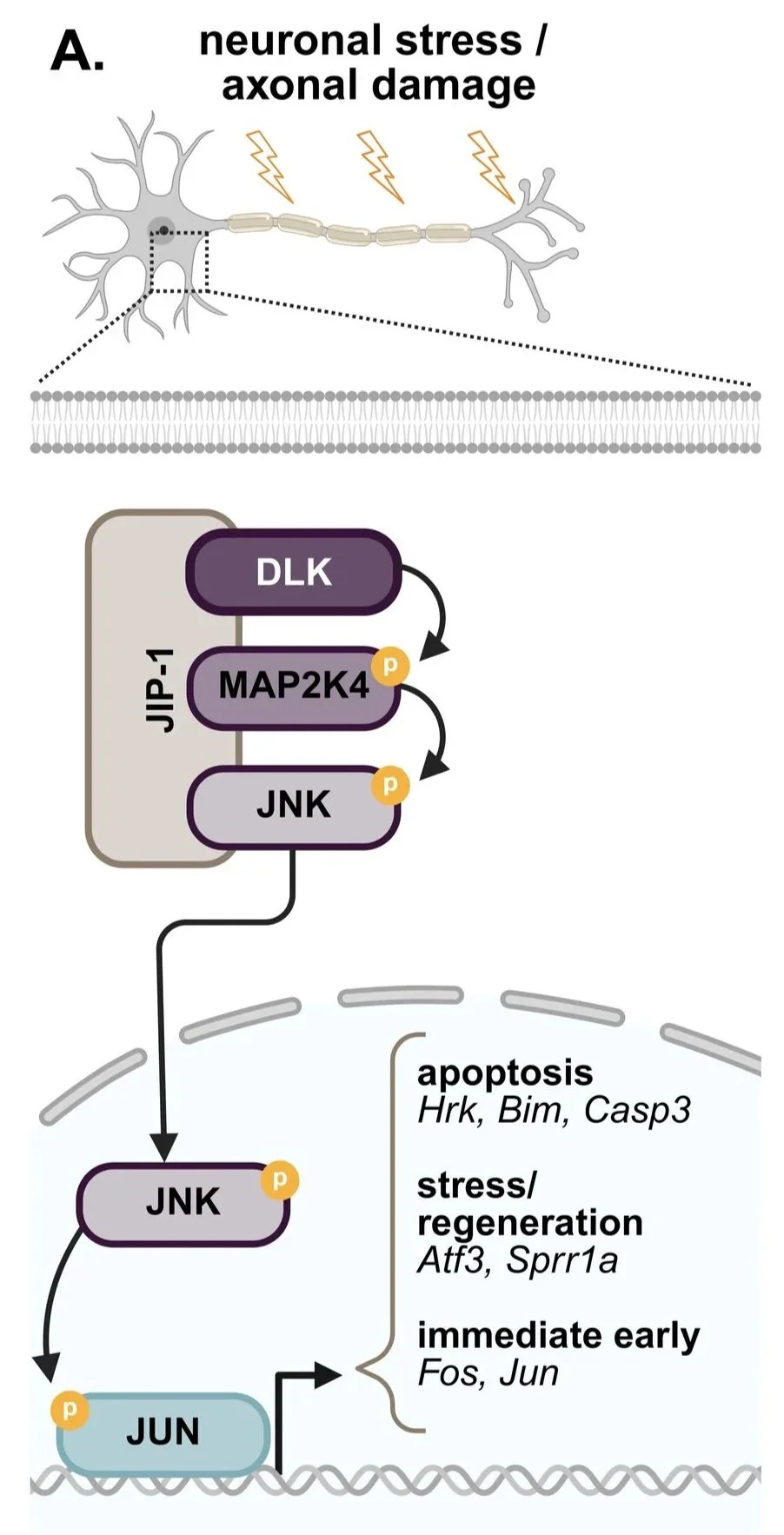
Figure 1A from the paper: Schematic of the DLK signaling cascade. Extracellular stress activates DLK, which phosphorylates MAP2K4, leading to phosphorylation of JNK. JNK translocates to the nucleus and activates the transcription factor JUN.
Our latest research, “DLK inhibition has sex-specific effects on neuroprotection and locomotor recovery after spinal cord injury” (pdf here), focused on a protein called Dual Leucine Zipper Kinase (DLK). In other models of neurodegeneration, like glaucoma or Alzheimer’s, DLK acts as a primary stress sensor. When a neuron is damaged, DLK sends a "death signal" through the JNK pathway (a molecular messenger system), telling the cell to shut down (Fig. 1 from the paper). We hypothesized that if we could block DLK and this death signal immediately after a spinal cord injury, we could shield these vulnerable neurons and help mice regain more movement.
To test this, we used a selective DLK inhibitor (IACS-52825) in a mouse model of spinal cord contusion. This work was led by Dr. John Aldrich (a Research Scientist in the lab), in collaboration with Dr. W. Jim Ray and other experts at the Belfer Neurodegeneration Consortium at the University of Texas MD Anderson Cancer Center. Using single-nucleus RNA sequencing, we confirmed that the DLK pathway is activated in neurons for at least a week following injury. In addition, the drug effectively entered the spinal cord and successfully suppressed the stress-activated signals.
However, the functional results took us by surprise.
A Surprising Sex Difference
Interestingly, our results contradicted our prediction. Instead of improving recovery, inhibiting DLK made outcomes worse after SCI - but only in males.
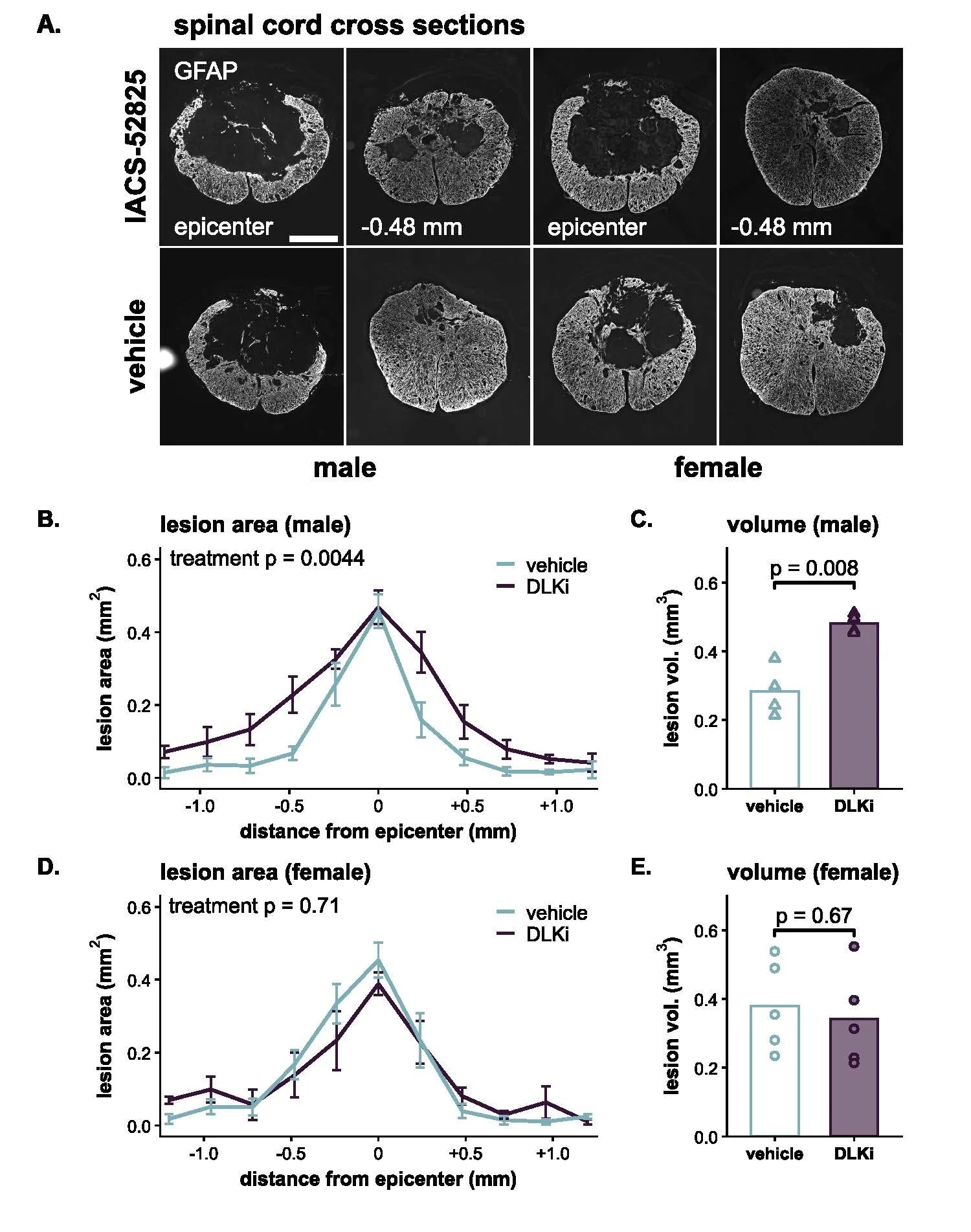
Figure 6 from the paper: DLK inhibitor increased lesion size in males, but not females after SCI. (A) In males (left), treatment with the DLK inhibitor led to significantly larger lesion areas (the central gap) compared to controls. (B,C) Quantification of lesion area and volume in males and females.
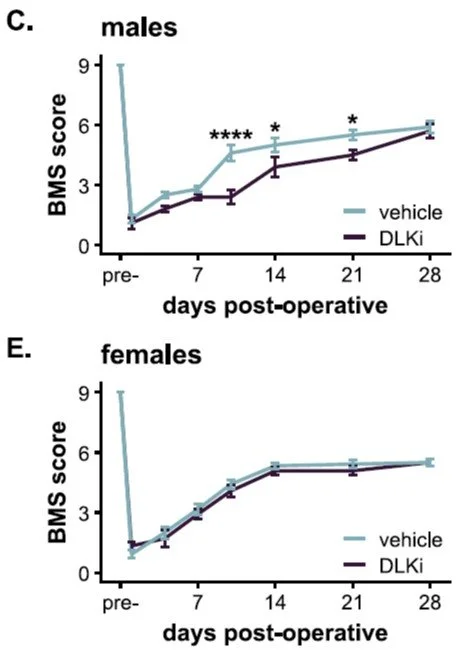
As shown in Figure 6, male mice treated with the inhibitor had 71% larger SCI lesions. This worsened damage was associated with a delay in their ability to regain coordinated walking. The female mice told a completely different story; the drug had no significant effect on their lesion size or their locomotor recovery after SCI (as seen in Figure 5).
Figure 5C and E from the paper: DLK inhibitor worsened open field locomotor recovery in males, but not females after SCI.
IMPLICATIONS
These results underscore why we must study both sexes in medical research. In the male spinal cord, the DLK "stress signal" might be beneficial in the early days post-injury - perhaps helping to coordinate the initial immune response or priming neurons for repair. By blocking DLK, we may have inadvertently disrupted a necessary part of the body's natural repair process. Further, broad inhibition of DLK likely impacts various cell types, including both neurons and oligodendrocytes. DLK also likely performs different functions in different parts of the cell. Optimizing how we target DLK - considering specific cell types, locations, and timing - could still lead to effective therapies for SCI and other neurologic conditions.
Check out the full study here: “DLK inhibition has sex-specific effects on neuroprotection and locomotor recovery after spinal cord injury” (pdf)
Authors (Gaudet lab members bolded):
John C. Aldrich, Samantha M. Alman, Sydney E. Lee, Ashley R. Scheinfeld, Chelsea C. Zhang, Averi L. Pike, Fiona C. Bremner, Olivia Calderon, Sunil Goodwani, William J. Ray, Andrew D. Gaudet